
Self-Association and Nitroaromatic-Induced Deaggregation of Pyrene Substituted Pyridine Amides | Journal of the American Chemical Society

Crystallography and QM/MM Simulations Identify Preferential Binding of Hydrolyzed Carbapenem and Penem Antibiotics to the L1 Metallo-β-Lactamase in the Imine Form | Journal of Chemical Information and Modeling

Multiscale Workflow for Modeling Ligand Complexes of Zinc Metalloproteins | Journal of Chemical Information and Modeling

Self-Association and Nitroaromatic-Induced Deaggregation of Pyrene Substituted Pyridine Amides | Journal of the American Chemical Society

Trimesic acid–Theophylline and Isopthalic acid–Caffeine Cocrystals: Synthesis, Characterization, Solubility, Molecular Docking, and Antimicrobial Activity | Crystal Growth & Design

Cocrystals of Oxymatrine: Reducing Hygroscopicity and Affecting the Dissolution Rate | Crystal Growth & Design
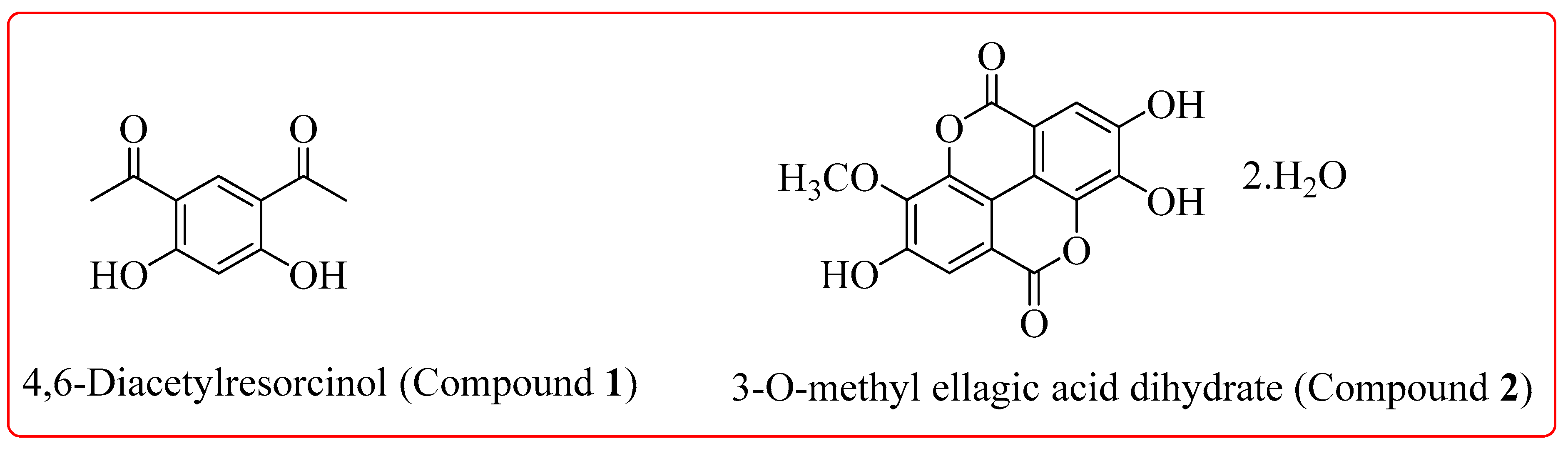
Crystals | Free Full-Text | Isolation, Identification, Spectral Studies and X-ray Crystal Structures of Two Compounds from Bixa orellana, DFT Calculations and DNA Binding Studies

Co-Crystals of Sulfamethazine with Some Carboxylic Acids and Amides: Co-Former Assisted Tautomerism in an Active Pharmaceutical Ingredient and Hydrogen Bond Competition Study | Crystal Growth & Design











